Real-world Evidence Assessments and Needs Guidance (REAdi) Tool
A Framework for Evaluating Real-World Evidence
The Real-world Evidence Assessments and Needs Guidance (REAdi) Tool is a web-based tool to help healthcare stakeholders and decision makers assess existing real-world evidence (RWE) in health technology assessment (HTA) processes.
REAdi was developed by investigators at The CHOICE Institute. It was partially funded through an unrestricted gift to the University of Washington from a consortium of eleven biomedical companies.
The REAdi framework is comprehensive in leading the user through the decision making process in five phases:
Phase 1: User defines research question in the Population, Intervention, Comparator, Outcome(s), Timing, Study Design/ Setting (PICOTS) format.
Once defined, REAdi automatically synthesizes terms, creates and executes a PubMed search strategy. Citations of relevant studies are returned for review.
Phase 2: REAdi guides users to a validated, publically available, embedded, quality-rating tool tailored to the design of each study.
Phase 3: User is prompted to rate the strength of the body of evidence for each outcome using the publically available GRADEPro (Grading of Recommendations, Assessment, Development and Evaluations) criteria.
Phase 4: Questions are posed to assess applicability and sufficiency of evidence to inform HTA adoption decisions; and to facilitate coverage decisions relevant to the immediate payer decision need at hand.
Phase 5: Summary results are presented in a tabular and graphical report.
REAdi is presented through an easy-to-use R Shiny interface, using drop-down menus, branching logic, and piping. The tool also harbors the functionality to print screen and save literature reviews allowing one to work on multiple projects simultaneously.
R Shiny app developed by:
- Brennan Beal (CHOICE Graduate Student Research Assistant)
- Stanley Wang (UW School of Pharmacy PharmD student)
CHOICE Faculty and Student Participants:
Beth Devine (Principal Investigator; CHOICE faculty)
Shuxian Chen (CHOICE Graduate Student Research Assistant)
Jamie Ta (CHOICE alumna)
Aasthaa Bansal (CHOICE faculty)
Joshua Carlson (CHOICE faculty)
David Veenstra (CHOICE faculty)
Anirban Basu (CHOICE faculty)
The investigators gratefully acknowledge the assistance of:
Doug Barthold (CHOICE faculty)
Jennifer Graff, National Pharmaceutical Council
Sophia Yun and other colleagues at Premera Blue-Cross
Diana Louden, UW Health Sciences Librarian
Joanne Rich, UW Health Sciences Librarian
Tom Hopkins (CHOICE alumnae)
Ashley Kim (CHOICE alumnae)
Eddie Neuberger (CHOICE alumnae)
Attendees at the invited workshop delivered at the 2018 AMCP Nexus Conference, Orlando, FL
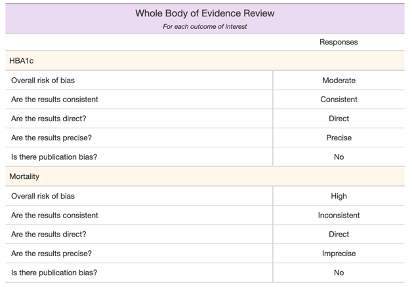
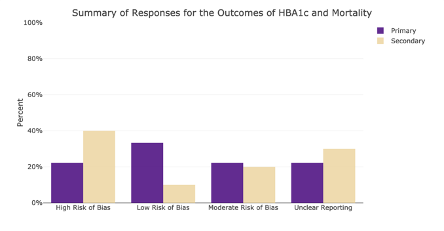
Example Output from Phase 5:
1. An Interactive bar plot to summarize one’s responses from their individual literature evaluation, which relies on curated tools specific to a given type of study
2. A table that summarizes the entire body of evidence, based on the reviewer’s responses (and manual input).