UW-AbbVie Fellowship
HEOR FELLOWSHIP PROGRAM 2026-2028
Contents
HEOR FELLOWSHIP PROGRAM 2026-2028
About AbbVie
At AbbVie, we strive to make a remarkable impact on the lives of patients. Our focus is clear – we are here to serve patients and we are committed to doing right by them.
We are a global, research-based biopharmaceutical company committed to discovering, developing and delivering innovative new medicines with distinct and compelling benefits for people.
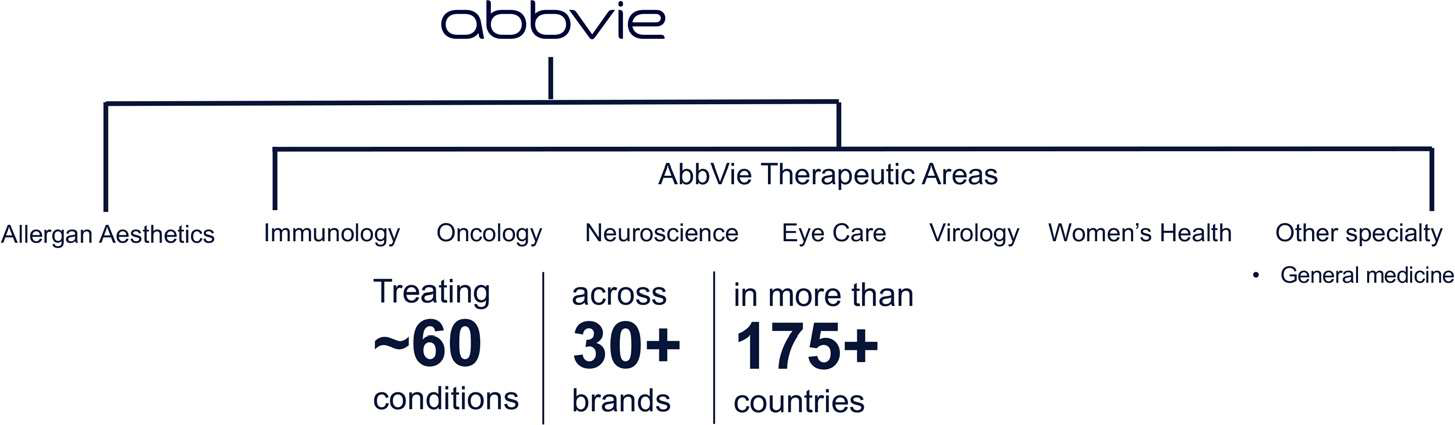
We are a passionate, diverse and inclusive organization with a culture that supports the best ideas, wherever they originate. AbbVie therapeutic areas include the following: immunology, oncology, neuroscience, eye care, virology, women’s health, other specialty. Treating ~60 conditions, across 30+ brands in more than 175+ countries.
 We bring people together because we know that collaboration is the key to breaking barriers and exploring new frontiers in science. We take smart risks that lead to transformative breakthroughs and can change lives for people all over the globe.
We bring people together because we know that collaboration is the key to breaking barriers and exploring new frontiers in science. We take smart risks that lead to transformative breakthroughs and can change lives for people all over the globe.
Our commitment to making a remarkable impact doesn’t end at developing medicines; it begins there. We provide broad support to our patients and help address the health needs of underserved communities. We strive to protect our environment and to make a positive impact in the areas where we live and work.
About the University of Washington
The University of Washington is a world-class leader in health sciences education and has been consistently listed in US News and World Report as one of the top 10 institutions for its graduate programs. The University is particularly noted for its strong research programs and since 1974 has received more federal funds for research and training than any other public university in the United States. It fosters a highly collegial atmosphere between departments and academic units, creating a particularly attractive environment for such partnerships between the public and private sectors.
The Comparative Health Outcomes, Policy, and Economics (CHOICE) Institute is housed within the University of Washington School of Pharmacy, which boasts nearly 125 years of academic and research success. Faculty in the program (1) provide advanced research training for graduate students, postdoctoral fellows and clinicians (PharmDs, MDs), (2) conduct rigorous scientific research in economic evaluations and outcomes of pharmaceuticals and other health technologies, and (3) translate and disseminate the results of research into clinical practice and healthcare policy, with the goal of improving patient care.
The CHOICE Institute faculty participating in the fellowship have been chosen both for their dedication to teaching and their excellence in research. Each is trained either as a health economist or health services researcher and all have an extensive history of effective economic evaluations and pharmacoepidemiologic research.

Noemi Kreif, MSc, MA, PhD
Assistant Professor of Health Economics
University of Washington
nkreif@uw.edu

Kyu Lee, PhD
Endowed Assistant Professor of Health Decision Sciences
University of Washington
kyueunl@uw.edu

Jae Lee, PharmD, MS
Associate Director, HEOR
AbbVie
jaehyun.lee@abbvie.com
First-Year Fellows

Andrew Cotterill, PharmD
Andrew is a PharmD graduate from Nova Southeastern University. His interest in HEOR developed during his pharmacoepidemiology research experiences, where he leveraged his computer science and programming background to synthesize data-driven evidence and model drug utilization with the goal of identifying treatment gaps to better inform patient care strategies. Under the guidance of the experts at CHOICE and AbbVie, Andrew is now focused on further developing his skillset and wants to explore the use of machine learning and AI in real-world evidence generation to improve the impact HEOR has on healthcare outcomes.

Janice Jang, PharmD
Janice is an incoming first-year UW CHOICE-AbbVie HEOR fellow and is a recent PharmD graduate of the University of Arizona. She developed an interest in HEOR through various research experiences including her summer research internship at the University of Arizona on the cost-efficiency analysis of adalimumab and its biosimilars. She looks forward to enhancing her skill sets in data analysis, economic modeling, and real-world evidence generation and is motivated to improve patient access and health outcomes. Janice is eager to learn from the experts at the CHOICE Institute and AbbVie.
Second-Year Fellows

Rachel Kneitel, PharmD, MS
Rachel earned her PharmD from St. John’s University. Her pharmaceutical industry internship and rotational experiences in HEOR, Market Access Analytics, and pharmacoeconomic research evaluating antibody-drug conjugates during pharmacy school shaped her interest in HEOR. These experiences have allowed her to design compelling value stories by linking real-world data to draw valuable insights into healthcare practices and capture the patient journey. In her second year, Rachel will work with Yanqing Xu and the hematology team across their therapeutic portfolio, spanning commercial and late-stage assets. Rachel is excited to build upon her first-year fellowship experience and leverage these skills in upcoming projects, further strengthening her foundation in HEOR.
Abstracts from Fellowship:
- Kneitel R, Xu Y, Kreif N. Strategies in Post-Transplant Maintenance Therapy for Acute Myeloid Leukemia: A Systematic Review. UW Corporate Advisory Board, September 2024, Seattle, WA.
- Kneitel R, Kreif N. Cost-Effectiveness of Xanomeline and Trospium Chloride for Schizophrenia in the United States. ISPOR, May 2025, Montreal, CA.
Manuscript from Fellowship:
- Kneitel R, Kreif N. Healthcare Resource Utilization and Costs in Patients with AML Treated with Post-Transplant Maintenance Therapy. (In Progress)

Olivia Yip, PharmD, MS
Olivia is a second-year UW/AbbVie HEOR Postdoctoral Fellow. She received her PharmD from the University of California, San Diego, where she developed a strong interest in HEOR through a variety of research experiences and externships. In her first year, Olivia developed a cost-utility model to assess the clinical outcomes and economic impact of expanding access to oseltamivir by making it available without a prescription in the U.S. healthcare system. She also contributed to studies examining neuropsychiatric symptoms in patients with Alzheimer’s disease, including a literature review on the prevalence of depression, anxiety, and apathy, as well as an analysis of healthcare utilization and cost burden associated with depression in this population. In her second year, Olivia is supporting HEOR strategy and evidence generation for assets in Parkinson’s disease. She is excited to continue building her skills and contribute to meaningful healthcare decision-making alongside mentors at CHOICE and AbbVie.
Abstracts from Fellowship:
- Yip O, Lee K. Cost-Utility Analysis of Non-Prescription Oseltamivir for Influenza Treatment: U.S. Healthcare Sector and Limited Societal Perspective. Presented at: ISPOR—The Professional Society for Health Economics and Outcomes Research Annual Meeting. May 2025; Montreal, Canada.
- Yip O, Sepassi A. Evaluation of Healthcare Expenditures and Resource Utilization in People with Cancer. Presented at: Western PharmacoEconomics Conference (WPEC). March 2025; Seattle, WA.
- Yip O, Lee K. Cost-Utility Analysis of Non-Prescription Oseltamivir for Influenza Treatment: U.S. Healthcare Sector Perspective. Presented at: Western PharmacoEconomics Conference (WPEC). March 2025; Seattle, WA.
- Yip O, Lee J, Lee K. Prevalence of Depression, Anxiety, and Apathy in Patients with Alzheimer’s Disease. Presented at: Corporate Advisory Board Meeting. September 2024; Seattle, WA.
Manuscripts from Fellowship:
- Yip O, English C, Lee K. Healthcare Utilization and Cost Burden Associated with Depression among Patients with Alzheimer’s Disease: A Retrospective Cohort Analysis. (In Progress)
- Yip O, Lee K. Evaluating the Value of Oseltamivir Reclassification for Influenza: A Cost-Effectiveness Analysis from U.S. Healthcare Sector and Limited Societal Perspectives. (In Progress)
Past Fellows
For more details on previous graduates, including their current positions of employment, please visit Alumni Graduate Students & Fellows
The University of Washington/AbbVie HEOR Fellowship is a two-year program that provides training and hands-on experience in a broad-range of health economics and outcomes research topics. The program is structured to allow fellows to gain proficiency in health outcomes strategy and research as conducted in both academic and pharmaceutical industry settings.
The Fellowship Program
 Fellows will spend their first year at the University of Washington (Seattle, WA), and the second year at AbbVie (Irvine, CA; Madison, NJ; or North Chicago, IL). The objective of the first year is to educate fellows on the principles and methods used in health economics and outcomes research. During the second year, fellows are provided with opportunities to gain practical knowledge and experience with the use of clinical, economic, and humanistic data by supporting the development and commercialization of pharmaceutical products. This will be accomplished by participating in work activities undertaken by the HEOR team.
Fellows will spend their first year at the University of Washington (Seattle, WA), and the second year at AbbVie (Irvine, CA; Madison, NJ; or North Chicago, IL). The objective of the first year is to educate fellows on the principles and methods used in health economics and outcomes research. During the second year, fellows are provided with opportunities to gain practical knowledge and experience with the use of clinical, economic, and humanistic data by supporting the development and commercialization of pharmaceutical products. This will be accomplished by participating in work activities undertaken by the HEOR team.
Compensation includes a competitive stipend, basic medical insurance, conference attendance, relocation from Seattle to Irvine, and holiday/vacation time. A Master’s (MS) Degree is awarded upon completion of all course requirements and thesis project.
Fellowship Structure
After completion of the first year, the fellow will have a clear understanding of:
- General principles of economics and in particular how economics influences
the use, cost, and availability of pharmaceuticals.
- The complex structure of drug policy and how it determines relationships between consumers, health care providers, insurers, payers, and government agencies.
- The breadth and scope of analytical methods needed for the assessment
of Patient Reported Outcomes (PROs), economic modeling, retrospective database studies, and observational studies.
- Formulary development, maintenance and evaluation in institutional, managed care, and state-level government programs.
First Year Objectives
The objective of the first year is to provide the fellow with the research tools necessary for economic evaluation and policy assessment. The fellow will be given an overview of pharmaceutical economics and general health policy, and taught methods of economic assessment, health services research, epidemiology, clinical research design, and biostatistical analyses.
The fellow will be assigned a MS thesis project which is selected based on input from both AbbVie as well as the University of Washington mentors. Fellows are highly encouraged to complete their MS project during their first year. Additionally, fellows will also engage in ongoing projects with AbbVie throughout their first year, applying skills from previous pharmacy training as well as integrating new techniques developed from University of Washington courses. Applying learning to solve real-world problems is a constant focus of the fellowship.
The fellow’s actual course of study will be developed on an individual basis with Drs. Kyu Lee & Noemi Kreif but will consist of courses in biostatistics, epidemiology, economic evaluations and outcomes research, and health and pharmaceutical policy.
 Second Year Objectives
Second Year Objectives
The second year of training will be at AbbVie with the HEOR department.
This part of the program is designed to allow the fellow to learn about the role of economic evaluations and outcomes research in the drug development and commercialization process within the industry setting. The research may be on an existing or experimental product for a disease state that is of interest to AbbVie.
In addition to completing individual research project(s), fellows will participate in ongoing economic evaluations, outcomes research, and other projects in collaboration with the HEOR team. These experiences will provide the fellow with opportunities to interact
with team members in other departments at AbbVie to learn how economic analyses and outcomes research are used throughout the pharmaceutical industry. Fellows will see firsthand how outcomes research is utilized to demonstrate the place of AbbVie products in treatment pathways and its value.
Application Requirements
- You must hold U.S. citizenship or permanent residency status to apply. Unfortunately, we are unable to sponsor non-immigrant visas for this program nor do international students on OPT qualify.
- Degree in pharmacy or health-related discipline.
- Previous research experience (preference will be given to those with a high academic standing).
- Excellent oral and written communication skills, ethics, professionalism, leadership and an interest in the biopharmaceutical industry.
- Curriculum vitae, cover letter, three letters of recommendation.
Selection of fellows will be made by a committee composed of University of Washington faculty members and representatives from the respective company.
Application Process Timeline:
- Eligibility: Degree in Pharmacy by July 1, 2026
- Application Opens: September 1, 2025
- Application Deadline: Monday, October 20, 2025
- Interview Notification: Monday, October 27, 2025
- Interviews: Week of November 3, 2025 (via Zoom)
- Offer Notification: Rolling, after interviews
- Program Start Date: July 1, 2026
How to Apply
- Login to the Graduate School application: Graduate School
- Select “Start Graduate Application”
- Complete all sections of the application along the left hand panel, starting with the Profile Information section.
- Under Program Selection, choose the following:
- Application type: Graduate
- Program: Pharmacy – Health Economics & Outcomes Research (MS)
- Review the Program Requirements and indicate the term and year you intend to enroll in the program
- Select Autumn 2025 (this is when the matriculation to the graduate program would begin).
- For the section, “Additional Materials”, download and complete the following Fellowship Selection Form.
- Upload the completed copy to your application.
- Continue with completing all other sections of the application, including Application Materials and Recommenders.
- Review your application and check for any missing required fields or errors. When ready, click Submit Application. Your application is not submitted until you have paid the application fee.
A more detailed instruction guide can be found in Application-Instructions.pdf.
Contact Information
For more information, please contact:
University of Washington
Marina Gano, M.Ed.
Graduate Program & Operations Manager
The CHOICE Institute, Department of Pharmacy
206-616-1383
uwsopchoice@uw.edu
AbbVie
Jae Lee, PharmD, MS
Associate Director, HEOR
2525 Dupont Drive
Irvine, CA 92612
(818) 726-7890
jaehyun.lee@abbvie.com